Consumer Guide: Drug Repository Program
The Drug Repository Program is a way for people who can't afford medicines to get what they need. Donors give medicine or medical supplies that they haven't used to participating pharmacies or medical facilities. These items then go to people with cancer or a long-term (chronic) condition. This page explains more about the program and how to get involved.
View the list of participating pharmacies and medical facilities in the accordions below.
- All Saints Cancer Center
- Phone: 262-687-5000
- Address: 3809 Spring St, Racine, WI 53405
- Contact person: David Fucile
- AltScripts
- Phone: 414-385-9500
- Address: 1636 Miller Park Way, West Milwaukee, WI 53214
- Contact person: Angelo Jones Pharm D.
- Ascension RX #1101
- Phone: 262-687-8430
- Address: 3807 Spring St, Racine, WI 53405
- Contact person: Molly Koser
- Ascension RX #1103
- Phone: 414-447-2585
- Address: 5000 W Chambers St, Milwaukee, WI 53210
- Contact person: Caroline Penne, Pharm D.
- Ascension RX #1104
- Phone: 262-780-4430
- Address: 19333 W North Ave, Brookfield, WI 53045
- Contact person: Uche Dike
- Ascension RX #1106
- Phone: 414-259-7440
- Address: 201 N Mayfair Road, Wauwatosa, WI 53226
- Contact person: Molly Koser
- Ascension RX #1107
- Phone: 414-647-5300
- Address: 3237 S 16th St, Room 121, Milwaukee, WI 53215
- Contact person: Jennifer York-Niemann
- Ascension RX #1109
- Phone: 262-243-6700
- Address: 13133 N Port Washington Road, Mequon, WI 53097
- Contact person: Michelle Hoff-Hilderbrand
- Ascension RX #1111
- Phone: 715-355-9577
- Address: 1506 S Oneida St, 1st Floor, Appleton, WI 54915
- Contact person: Thomas Vick
- Ascension RX #1112
- Phone: 414-585-1303
- Address: 2301 N Lake Dr, Milwaukee, WI 53211
- Contact person: Thomas Vick
- Ascension RX #1116
- Phone: 414-281-7500
- Address: 7400 W Rawson Ave Suite 140, Franklin, WI 53132
- Contact person: Roy Clemence
- Aspirus Kidney Care Wausau
- Phone: 715-847-0019
- Address: 3000 Westhill Drive, Suite 106, Wausau, WI 54401
- Contact person: Jamie Anderson
- Aspirus Pharmacy -Rhinelander
- Phone: 715-361-4770
- Address: 2251 Northshore Drive, Rhinelander, WI 54501
- Contact person: Allison Grafwallner
- Aspirus Pharmacy - Stanley
- Phone: 715-355-9573
- Address: 1120 Pine St, Stanley, WI 54768
- Contact person: Macy Porlier
- Aspirus Pharmacy - Stevens Point
- Phone: 715-346-0750
- Address: 900 Illinois Avenue, Stevens Point, WI 54481
- Contact person: Zachary Miller
- Aspirus Pharmacy - Westwood Wausau
- Phone: 715-355-9573
- Address: 1901 Westwood Center Blvd, Wausau, WI 54401
- Contact person: Macy Porlier
- Aurora Sinai Medical Center Outpatient Pharmacy
- Phone: 414-219-3100
- Address: 945 N 12th St, Milwaukee, WI 53233
- Contact person: Brian Johnson
- Bellin Memorial Hospital
- Phone: 920-433-3648
- Address: 744 South Webster, Green Bay, WI 54301
- Contact person: Laura Alar
- Bohlman Drugstore, Inc.
- Phone: 608-375-4466
- Address: 1028 Wisconsin Ave, Boscobel, WI 53805
- Contact person: Michelle Farrell
- Center Pharmacy
- Phone: 608-647-8918
- Address: 190 N Orange St, Suite 2, Richland Center, WI 53581
- Contact person: Michelle Farrell
- Church Health Services
- Phone: 920-887-1766
- Address: 115 N Center St, Beaver Dam, WI 53916
- Contact person: Dennis Breidenbach
- Community Pharmacy
- Phone: 608-251-4454
- Address: 130 S Fair Oaks, Madison, WI 53704
- Contact person: Aimee Speers
- Dennis L. Hunt Pharmacy, Inc. DBA Omro Pharmacy
- Phone: 920-685-5041
- Address: 109 E Main St, Omro, WI 54963
- Contact person: Kenneth A. Bressers
- Evergreen Pharmacy
- Phone: 414-533-6600
- Address: 10101 W Greenfield Ave, Suite 130, West Allis, WI 53214
- Contact person: Andrew Hochradel, Pharm D
- Family Health La Clinica Wautoma Pharmacy
- Phone: 800-942-5330
- Address: 400 S. Townline Road, Wautoma, WI 54982
- Contact person: Michael Peterson
- First Presbyterian Church - Wausau Free Clinic
- Phone: 715-355-9630
- Address: 360 Grand Ave, Suite 600, Wausau, WI 54403
- Contact person: Zachary Miller
- Fitchburg Family Pharmacy
- Phone: 608-886-7117
- Address: 1890-F13050 Cahill Main #6, Fitchburg, WI 53711
- Contact person: Thad Schumacher
- Froedtert Pharmacy #025
- Phone: 414-805-6500
- Address: 8800 W Doyne Ave, Milwaukee, WI 53226
- Contact person: Khushboo Modi
- Froedtert Pharmacy #050
- Phone: 414-805-6500
- Address: 8850 William Coffey Dr, Milwaukee, WI 53226
- Contact person: Khushboo Modi
- Froedtert Pharmacy #075
- Phone: 414-805-6500
- Address: 900 N 92nd St, Milwaukee, WI 53226
- Contact person: Khushboo Modi
- Good Value Pharmacy
- Phone: 262-697-5744
- Address: 3207 80th St, Suite 100, Kenosha, WI 53142
- Contact person: Scott Christofferson
- Good Value Pharmacy Racine
- Phone: 262-632-6561
- Address: 5220 Washington Ave, Suite #101, Racine, WI 53406
- Contact person: Brian Bock
- Good Value Pharmacy/Larson - Mayer Pharmacy
- Phone: 262-658-8124
- Address: 3825 39th Ave, #100, Kenosha, WI 53144
- Contact person: Andrea Wood
- Gundersen Lutheran East Pharmacy
- Phone: 608-775-1000
- Address: 724 Denton St, La Crosse, WI 54601
- Contact person: Jim Lukas
- Gwidt Pharmacy
- Phone: 715-253-2164
- Address: 203 E Westgor Ave, Wittenberg, WI 54499
- Contact person: Katie Hanke
- Health connections Incorporated
- Phone: 414-999-1099
- Address: 4655 N Port Washington Road, Glendale, WI 53212
- Contact person: Erica Sinclair
- Healthnet of Rock County, Inc.
- Phone: 608-756-4638
- Address: 113 S Franklin St, Janesville, WI 53548
- Contact person: Keisha Owens
- Hope Clinic and Care Center
- Phone: 920-931-1150
- Address: 1814 Appleton Road, Menasha, WI 54952
- Contact person: Dr. Robert Makeever
- Lake Area Free Clinic
- Phone: 262-569-4990
- Address: 856 B Armour Road, Oconomowoc, WI 53066
- Contact person: Mary Reich
- Marshfield Clinic Pharmacy Central Avenue
- Phone: 715-389-5900
- Address: 630 South Central Avenue, Suite #106, Marshfield, WI 54449
- Contact person: Rob Nichols
- Marshfield Clinic Pharmacy Chippewa Center
- Phone: 715-726-4177
- Address: 2655 County Hwy I, Chippewa Falls, WI 54729
- Contact person: Steve Bartig
- Marshfield Clinic Pharmacy Eau Claire Center
- Phone: 715-858-4811
- Address: 2116 Craig Road, Eau Claire, WI 54701
- Contact person: Bruce Bergmann
- Marshfield Clinic Pharmacy Ladysmith Center
- Phone: 715-532-2323
- Address: 906 College Avenue West, Ladysmith, WI 54848
- Contact person: Jim Rubin
- Marshfield Clinic Pharmacy Minocqua Center
- Phone: 715-358-1216
- Address: 9576 Hwy 70 STE A, Minocqua, WI 54548
- Contact person: Alex Otte
- Marshfield Clinic Pharmacy Mosinee Center
- Phone: 715-693-9181
- Address: 390 Orbiting Dr, Mosinee, WI 54455
- Contact person: Holly Dillenbeck
- Marshfield Clinic Pharmacy Oakwood Center
- Phone: 715-858-4224
- Address: 3501 Golf Road, Eau Claire, WI 54701
- Contact person: Dana Whittlinger
- Marshfield Clinic Pharmacy Wausau Center
- Phone: 715-847-3302
- Address: 2727 Plaza Drive, Wausau, WI 54401
- Contact person: Eric Paulson
- Marshfield Clinic Pharmacy- First Floor
- Phone: 715-387-9100
- Address: 1000 N Oak Avenue, Marshfield, WI 54449
- Contact person: Adam Maguire
- Marshfield Clinic Pharmacy-Rice Lake Center
- Phone: 715-236-8103
- Address: 1700 West Stout St, Rice Lake, WI 54868
- Contact person: Marni Britton
- Marshfield Clinic Pharmacy-Specialty Pharmacy
- Phone: 715-221-8842
- Address: 1000 N Oak Avenue, Marshfield, WI 54449
- Contact person: Holly Fayas
- Marshfield Clinic Remote Dispensing Mercer
- Phone: 715-358-1217
- Address: 5110 N Hwy 51, Mercer, WI 54547
- Contact person: Alex Otte
- Marshfield Clinic Remote Dispensing Merrill Center
- Phone: 715-539-0118
- Address: 1205 O'Day St, Merrill, WI 54452
- Contact person: Alex Otte
- Marshfield Clinic Remote Dispensing Riverview Center
- Phone: 715-858-4366
- Address: 1000 Starr Avenue, Eau Claire, WI 54703
- Contact person: Steve Bartig
- Marshfield Clinic Remote Dispensing Weston Center
- Phone: 715-393-1040
- Address: 3501 Cranberry Blvd, Weston, WI 54476
- Contact person: Eric Paulson
- Mayo Clinic Health System Eau Claire
- Phone: 715-464-8144
- Address: 1400 Bellinger St, Eau Claire, WI 54703
- Contact person: Kaitlyn Bailey
- Mayo Clinic Health System Skemp Pharmacy
- Phone: 608-392-7646
- Address: 800 West Ave. S, La Crosse, WI 54601
- Contact person: Grant Perschke
- Mayo Clinic Health System Northland Pharmacy
- Phone: 715-233-4703
- Address: 1222 E Woodland, Barron, WI 54812
- Contact person: Sheila Lee
- Mayo Clinic Health System Red Cedar Pharmacy
- Phone: 715-233-4703
- Address: 2321 Stout Road, Menomonie, WI 54751
- Contact person: Sheila Lee
- Open Arms Free Clinic
- Phone: 262-379-1401
- Address: 205 E Commerce Ct, Elkhorn, WI 53121
- Contact person: William Clark
- Pharmacy at Froedtert West Bend Hospital
- Phone: 414-805-6500
- Address: 3200 Pleasant Valley Road, West Bend, WI 53095
- Contact person: Khushboo Modi
- Pick N Save Pharmacy
- Phone: 262-253-4705
- Address: N95 W18273 County Line Road, Menomonee Falls, WI 53051
- Contact person: Gary Miller
- Reedsburg Area Medical Center
- Phone: 608-524-6177
- Address: 2000 N Dewey Avenue, Reedsburg, WI 53959
- Contact person: Matt Huppert
- Rhinelander Hometown Pharmacy
- Phone: 715-420-1685
- Address: 345 Brown St, Rhinelander, WI 54501
- Contact person: David W. Schick
- Sage Specialty Pharmacy
- Phone: 414-861-7243
- Address: 4001 N Oakland Ave, Shorewood, WI 53211
- Contact person: Jon S. Phillips
- Salvation Army Red Shield Medical Clinic
- Phone: 920-684-7117
- Address: 411 N 6th St, Manitowoc, WI 54220
- Contact person: Mary Ellen Beebe
- Saturday Clinic for Uninsured
- Phone: 414-955-2641
- Address: 1121 E North Ave, Milwaukee, WI 53212
- Contact person: Dr. Rachele Harrison
- Sixteen Street Community Health
- Phone: 414-672-1353
- Address: 1032 South Cesar Chavez Dr, Milwaukee, WI 53204
- Contact person: Annaliza Torres APNP
- Smith Pharmacy
- Phone: 920-788-8888
- Address: 1800 Freedom Road, Suite D, Little Chute, WI 54140
- Contact person: Nic Smith
- St. Clare Health Mission of Monroe County
- Phone: 608-366-5343
- Address: 310 W Main St, Sparta, WI 54656
- Contact person: Dr. Michael Saunders
- St. Vincent de Paul Charitable Pharmacy
- Phone: 608-268-0355
- Address: 2033 Fish Hatchery Road, Madison, WI 53725-9686
- Contact person: Yolanda Tolson
- St. Vincent de Paul Free Medical Clinic
- Phone: 715-387-0395
- Address: 149 N Central Ave, Marshfield, WI 54449
- Contact person: Alexia Knick
- Stevens Point Hometown Pharmacy
- Phone: 715-697-0780
- Address: 3423 Main St, Suite A & B, Stevens Point, WI 54481
- Contact person: Tessa Garr
- Tomah Health
- Phone: 608-377-8409
- Address: 501 Gopher Drive, Tomah, WI 54660
- Contact person: Melissa Duoss
- Tomahawk Pharmacy
- Phone: 715-458-6600
- Address: 844 N 4th St, Tomahawk, WI 54487
- Contact person: Tyler Stevenson
- UW Health Eastpark Medical Center Pharmacy
- Phone: 608-916-1400
- Address: 4621 Eastpark Blvd, Room C1142, Madison, WI 53718
- Contact person: Cody Moriearty
- UW Health Pharmacy Services
- Phone: 608-263-1280
- Address: 600 Highland Ave, Madison, WI 53792
- Contact person: Cody Moriearty
- Vivent Pharmacy
- Phone: 414-223-6820
- Address: 820 N Plankinton, Milwaukee, WI 53203
- Contact person: Nicole Lentz
- Vivent Pharmacy
- Phone: 608-316-8612
- Address: 600 Williamson St, Madison, WI 53703
- Contact person: Ann Schneps-Visor
- White Cross Pharmacy
- Phone: 715-561-5666
- Address: 314 Silver St, Hurley, WI 54534
- Contact person: Andrew Heckel
- Wisconsin Fertility Institute
- Phone: 608-824-0075
- Address: 3146 Deming Way, Middleton, WI 53562
- Contact person: Dr. Gretchen Collins
Partial participation means the facility only will accept donations and transfer to a facility that is a full participant.
- Agnesian Pharmacy Plus
- Phone: 920-926-8723
- Address: 420 E Division St, Fond du Lac, WI 54935
- Contact person: Nancy Hyland
- Froedtert Pharmacy #100
- Phone: 262-836-5411
- Address: 3200 Pleasant Valley Road, Suite 1A, West Bend, WI 53095
- Contact person: Mindy Eichinger
- Froedtert Pharmacy #150
- Phone: 262-532-3050
- Address: W180 N8000 Town Hall Road, Menomonee Falls, WI 53051
- Contact person: Katie Laufenberg
- Froedtert Pharmacy #175
- Phone: 414-955-3350
- Address: 1155 N Mayfair Road, Wauwatosa, WI 53226
- Contact person: Kevin Stutt
- Froedtert Pharmacy #200
- Phone: 414-805-6500
- Address: 925 N 87th St, Milwaukee, WI 53226
- Contact person: Khushboo Modi
- Froedtert Pharmacy #225
- Phone: 414-805-1295
- Address: 8900 W Doyne Ave, Milwaukee, WI 53226
- Contact person: Holly Ferrel
- Froedtert Pharmacy #325
- Phone: 414-346-8050
- Address: 7901 S 6th St, Oak Creek , WI 53154
- Contact person: Amy Calteux
- Froedtert Pharmacy #400
- Phone: 414-454-8150
- Address: 10000 W Bluemound, Wauwatosa, WI 53226
- Contact person: Kevin Lewis
- Froedtert Pharmacy #375
- Phone: 920-320-4400
- Address: 1650 S 41st St, Manitowoc, WI 54220
- Contact person: Holly Dewane
- Froedtert Pharmacy at Moorland Reserve Health Center
- Phone: 262-798-7386
- Address: 4805 S Moorland Road, Room G0502A, New Berlin, WI 53151
- Contact person: Nixheng-Yeng Xiong
- Froedtert Remote Dispensing Site #300
- Phone: 262-532-3050
- Address: 11430 N Port Washington Road, Mequon, WI 53092
- Contact person: Katie Laufenberg
- Gundersen La Crosse Clinic Pharmacy
- Phone: 608-775-5595
- Address: 1836 South Ave, La Crosse, WI 54601
- Contact person: James Bergkamp
- Gundersen Onalaska Clinic Pharmacy
- Phone: 608-775-8699
- Address: 3111 Gundersen Dr, Onalaska, WI 54650
- Contact person: Mary C. Erickson
- Pharmacy at Froedtert Menomonee Falls Hospital
- Phone: 262-257-3073
- Address: W180N8085 Town Hall Road, Menomonee Falls, WI 53051
- Contact person: Janelle Schwartz
Helpful Drug Repository Program forms
Download and complete these forms based on your needs.
- Drug Repository Program Notice of Participation or Withdrawal, F-62643 (Word) - Pharmacies or medical facilities must fill out this form if they want to join or leave the program.
- Drug Repository Program: Recipient Record, F-62645 - Anyone who receives drugs or supplies as part of the program must complete this form.
- Drug Repository Program: Transfer Record, F-62645A (Word) - Pharmacies and medical facilities must use this form if they give donated drugs or supplies to another pharmacy or medical facility.
- Drug Repository Program: Donation Record, F-62645B (Word) - Anyone who donates drugs or supplies as part of the program must complete this form.
- Drug Repository Program: Destruction Record, F-62645C (Word) - Pharmacies and medical facilities must use this form if they destroy or discard any donated drugs or supplies.
Drug donation FAQs (frequently asked questions)
Expand a section to find the answer to the question.
These statements must be true to donate a prescription medicine as part of this program:
- The donation includes a signed Drug Repository Program: Donation Record, F-62645B (Word) from the donor or an authorized representative.
- The drug is not adulterated or misbranded.
- Adulterated means that the drug is either:
- Damaged.
- Stored the wrong way.
- Not safe to use based on something that happened to it.
- Misbranded means the drug had the wrong label from the start. This makes it not safe to use.
- Adulterated means that the drug is either:
- The drug will not expire until at least 90 days after the donation date.
- The drug is in its original, unopened, tamper-evident packaging.
- Tamper-evident packaging means the package is sealed.
- Other approved packaging includes:
- An inhaler sealed in a foil wrapper.
- Injectable drugs with the manufacture vial cap intact.
No, you cannot donate controlled substances, such as:
- Lorazepam
- Morphine
- Oxycodone
These are often used to treat pain, anxiety, and sleep issues. You can't return these to a pharmacy or medical facility based on federal law, 21 CFR 1317.35(d).
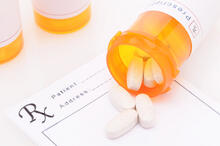
No, you can't donate drugs you get from the pharmacy in a bottle. This includes the common brown or amber bottles (see image for reference).
You can donate unused prescription medicines to any pharmacy or medical facility listed in this document:
Drug Repository Participants (PDF)
Contact a location to learn more about their specific hours and donation process.
Any person who lives in Wisconsin can receive drugs or supplies through the Drug Repository Program. The law sets priority of who gets the drugs or supplies first:
- Those who don't have insurance.
- Those who receive or qualify for Medicaid, Medicare, or other government-based health care.
- Everyone else who is otherwise eligible.
Learn more from Wis. Admin. Code § 148.07 (4).
You may be charged a fee at some pharmacies for the extra work they do when giving out donated drugs. This includes time spent:
- Counseling
- Documenting
- Preparing
The fee can't be more than $15.
Contact a participating pharmacy or medical facility to see which drugs they have. View Drug Repository Participants (PDF) for contact details.
Yes. Pharmacies and medical facilities can't give out drugs that have a restricted distribution status from the U.S. Food and Drug Administration.
If there's a question, the pharmacy or medical facility can call the drug maker directly or visit the U.S. Food and Drug Administration's website. Often, the maker of a restricted distribution drug has its own return policy for unused medicine.
It's important for all people in Wisconsin to have access to medicine and supplies they need to fight a disease.
State law (Wis. Stat. § 255.055) also authorized DHS to set up and maintain a drug repository program. DHS was required to come up with rules for the program. Wis. Admin. Code ch. DHS 148 summarizes these rules.
Who to contact for help
Questions about the Drug Repository Program? Contact douglas.englebert@dhs.wisconsin.gov.